Summary
My Master’s research thesis proposed a biocompatible, highly elastic, low cost device (modelled after a more complicated, costly drug delivery system) that aims to prevent tissue death by introducing stem cell therapies via hydrogel to the infarct region post-myocardial infarction and that ideally would also provide support to weakened areas of the left ventricle wall. The method of action of this device is to promote revascularization and tissue growth in the infarct region via stem cells, growth factors, and medication localization by hydrogel and patch implementation. I performed a series of tests to characterize the device.
Firstly, the tangential force of adhesion of the device under two different attachment techniques (glue and suture) was determined via Zwick force (pull-off) testing, under both unused and fatigued conditions. Fatigue was applied by applying strain to the device equal to maximum average ventricular displacement during diastole of heart disease patients. It was found the device’s integrity changed significantly up until the equivalent of six heartbeats, but not after this, so the strain was applied for six cycles. It was found that glue did not adequately attach the device, but suturing did, even when the device was fatigued.
Firstly, the tangential force of adhesion of the device under two different attachment techniques (glue and suture) was determined via Zwick force (pull-off) testing, under both unused and fatigued conditions. Fatigue was applied by applying strain to the device equal to maximum average ventricular displacement during diastole of heart disease patients. It was found the device’s integrity changed significantly up until the equivalent of six heartbeats, but not after this, so the strain was applied for six cycles. It was found that glue did not adequately attach the device, but suturing did, even when the device was fatigued.
Secondly, the burst pressure of devices with normal (0.0762 mm) and thicker (0.3038 mm) backing was determined to examine the ability of the device to withstand pressure when filling. It was determined that the device was acceptably resistant to pressure with both backings, but the effects of fatigue were less significant with the thicker backing, giving the device greater overall longevity.
Thirdly, the ability of the device to provide mechanical support to the weakened left ventricle was determined via a force-displacement study that compared the ability of a scar tissue silicone model to withstand strain without the device, with an empty device, and with a hydrogel-filled device (how it would likely exist in vivo). From this test it was determined that the hydrogel-filled device greatly enhanced the tissue’s mechanical strength by providing mechanical support during strain. This is likely due to the added stiffness of the filled device serving as a backbone to the displacing tissue.
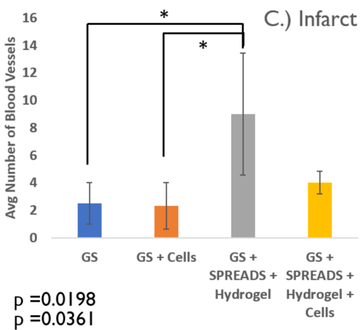
Finally, the extent to which revascularization was a method of action of the device was surmised by a histological analysis of a former pig study (Dolan et al.) done with a comparable device. This assessment quantified the success of the stem cell, medication, and hydrogel therapy delivered by the device to the infarct region by blind counting the number of blood vessels formed in the region. It was found that the device successfully delivered the therapy, which in turn was efficacious in revascularization in the infarct.
Thirdly, the ability of the device to provide mechanical support to the weakened left ventricle was determined via a force-displacement study that compared the ability of a scar tissue silicone model to withstand strain without the device, with an empty device, and with a hydrogel-filled device (how it would likely exist in vivo). From this test it was determined that the hydrogel-filled device greatly enhanced the tissue’s mechanical strength by providing mechanical support during strain. This is likely due to the added stiffness of the filled device serving as a backbone to the displacing tissue.
Finally, the extent to which revascularization was a method of action of the device was surmised by a histological analysis of a former pig study (Dolan et al.) done with a comparable device. This assessment quantified the success of the stem cell, medication, and hydrogel therapy delivered by the device to the infarct region by blind counting the number of blood vessels formed in the region. It was found that the device successfully delivered the therapy, which in turn was efficacious in revascularization in the infarct.
Overall, the therapeutic device was found to be most successful and durable when the thicker backing was employed, the device was sutured on, and when it was placed in the immediate infarct region due to therapy localization. In the future, the stem cell therapy may be optimized for even greater success and more in vivo testing must be completed to ensure the greatest efficacy for heart disease patients.
Main Tasks
- Device characterization (bench-top testing) and optimization
- Device manufacturing
- Tangential adhesion force testing
- Animal tissue handling
- Burst pressure testing
- Effects of fatigue
- Optimal device thickness and attachment method
- Analyze device's mechanical contribution to the left ventricle
- Comparative force study
- Force application; Hydrogel loading
- Thicker material provided more support
- Determine if revascularization is a method of action for remodeling prevention
- iPSC, Hydrogel, Medications (Global Standard (GS))
- Histological analysis (ImageJ) of a former pig study (Dolan et al.)
Skills Obtained
- Autodesk Inventor
- Device Burst Pressure Testing
- Histological Analysis
- ImageJ
- Medical Device Production
- Zwick Analysis
- Experiment Design and Execution
- Animal Tissue Handling
- Technical Writing/Thesis
- Hydrogel Handling
- Statistical Analysis
Significance
Heart disease affects people from all backgrounds and is the leading cause of death each year around the world. Myocardial infarction is the most prominent form of heart disease and occurs when ischemia to the left ventricle myocardial tissue results in cell necrosis in the region. The dead myocardial tissue is replaced by scar tissue, a phenomenon known as remodeling. This scar tissue exhibits different mechanical properties than that of the living tissue. The result is nonuniform muscle tissue and therefore weakened pumping ability from areas of less strength in the left ventricle (i.e., the scar tissue). Currently treatment for survivors of heart attacks is limited and does not restore healthy heart function, which may impede patients’ daily lifestyles. My Master’s research thesis proposed a biocompatible, highly elastic, low cost device (modelled after a more complicated, costly drug delivery system) that aims to prevent tissue death by introducing stem cell therapies via hydrogel to the infarct region post-myocardial infarction and that ideally would also provide support to weakened areas of the left ventricle wall. The method of action of this device is to promote revascularization and tissue growth in the infarct region via stem cells, growth factors, and medication localization by hydrogel and patch implementation.